Innovated Product
embed video
Quick Links
Contact Us

Twitter, Inc.
ADD: El-sarag towers No.2 Entrance No4Makram Ebid St.
Tel:02 22756545 - 02 26703616
E-mail:
info@mdcegy.com
Product Details
Symplicity Flex

What Is Treatment-Resistant Hypertension?
Treatment-resistant hypertension is blood pressure (BP) that remains above goal despite compliance with full doses of ≥3 antihypertensive medications of different classes; 1 of the 3 agents should be a diuretic and the treatment plan must include attention to lifestyle measures. Treatment-resistant hypertension could also include patients who achieve BP control but need ≥4 antihypertensive agents to do so.
Prevalence and Consequences
The exact prevalence of treatment-resistant hypertension is unknown.2 Small studies estimate it at approximately 5% in general practice and 50% or higher in nephrology clinics, depending on the severity of the underlying chronic renal disease. In the NHANES study, the estimated prevalence of treatment-resistant hypertension was 8.9% of all hypertensive adults and 12.8% of all drug-treated hypertensive adults in the US. Patients with treatment-resistant hypertension have been shown to have an approximately 3-fold increase in risk for cardiovascular (CV) events versus patients with controlled hypertension.5 No longitudinal study has specially evaluated the prognosis of resistant hypertension. The degree to which CV risk is reduced with treatment is unknown, but the benefits of successful treatment are likely substantial
Overview of the sympathetic nervous system and its role in hypertension
The Sympathetic Nervous System
.jpg)
Quantifying Human SNS Activity

Renal sympathetic nerves in cardiovascular diseases
Crosstalk Between Renal Nervous and CNS
.jpg)
Renin-Angiotensin-Aldosterone System (RAAS) in Hypertension

Increased central Sympathetic Drive in Hypertension

Chronic Effect of Increased Sympathetic Nerve Activity

Hypertension May Be Just the Begining
.jpg)
A Novel Therapy for Treatment-Resistant Hypertension
Medtronic offers a novel and innovative therapy for treatment-resistant hypertension. The SymplicityTM renal denervation system is a new, safe, and effective tool for healthcare professionals to use for patients who, until this point, have been unable to achieve target blood pressure levels despite multiple prescription medications, and are considered treatment-resistant. The Symplicity renal denervation system has been successfully used since 2007 to treat more than 4,000 patients with treatment-resistant hypertension worldwide. It was launched commercially in April 2010 and is currently available in parts of Europe, Asia, Africa, Australia and the Americas.

Manifold Set-up and Contrast Management
.jpg)
Symplicity™ Catheter Tip Features
.jpg)
Symplicity™ Catheter Handle Features

Guide Catheter Selection
- Key Guide Selection Criteria
– Takeoff angle of renal arteries
– Engagement at ostiumto prevent deep seating of guide catheter
– Soft tip at engagement with both active and balanced support

Guide Catheter Selection
.jpg)
Generator and Dispersive Electrode
- Generator
– Plug in RF generator and turn on switch on back panel
– Verify system beeps and all displays momentarily illuminate
– Press Continue
- Dispersive Electrode (Ground Pad)
– Place on the thigh or other non-bony area out of the angiogram field (shave if necessary for good contact)
– Ensure all other grounds are removed from patient

Single Patient Use Only
The Symplicity™ catheter is intended for single patient use
only – Do not resterilize or reuse. Reuse, reprocessing,
or resterilization may compromise device integrity and may
create the risk of transmission of infectious diseases from
one patient to another, which may result in injury, illness,
or death of the patient.
About the Procedure
Renal denervation is a novel therapy approved for treatment-resistant hypertension. The SymplicityTM renal denervation system consists of a generator and a flexible catheter. During this minimally invasive procedure, the interventionalist uses a steerable catheter with a radio frequency (RF) energy electrode tip. The RF energy is delivered to a renal artery via standard femoral artery access. A series of 2-minute ablations are delivered along each renal artery to disrupt the nerves

-
Equipment
– SymplicityTM catheter and generator
- Dispersive electrode (ground pad)– sent with Symplicity catheter
– 6 French renal guide catheter
(45-55 cm, RDN-D1/RDC-1, or IMA/LIMA) and introducer sheath
– Guide wire (0.014”, non-hydrophilic)
– Non-ionic contrast (dilute to 50:50)
– Heparinized saline flush bag (pressurized)
– Radiopaque ruler
-
Medications*
Common medications to have available for procedure
– Heparin
– Fentanyl/morphine or similar
– Midazolam
(eg, Dormicum, Versed) or similar
– Nitroglycerine
– Atropine Always treat distal to proximal
– Do not re-cross previously treated site

-
4-6 focal treatments
– 120 seconds per treatment
– ≥ 5 mm between locations
– Stable, unique locations
– Circumferential coverage
-
Common strategy (dependent on renal anatomy):
– Distal: Inferior and inferolateral locations
– Proximal: Superior and superolateral locations
– If positioning is unstable, avoid purely lateral treatments (possible electrode movement with respiration)
– Favor stable wall contact over circumferential treatment
-
PULL, ROTATE, ASSESS new location and prior treatment site with cine just prior to each treatment
– Delivering the SymplicityTM Catheter
-
Fully straighten tip of catheter – Never advance catheter while tip is deflected
– Do not cross recently treated sites
– Deliver Symplicity catheter through the renal guide catheter
.png)
Positioning the SymplicityTM Catheter
-
With electrode advanced to most distal treatment site, use handle lever to deflect catheter tip against vessel wall and retract to most distal treatment site

-
Good wall contact is important for the proper delivery of energy
– First deflect slightly and rotate to superior or inferior hemisphere
– Fully deflect tip to optimize electrode apposition to the vessel wall
– May not need to use handle lever to deflect tip in tortuous anatomy or proximal locations
Optimizing Treatment Sites – Impedance
-
Impedance may be used to confirm stable wall contact:
– Higher impedance may indicate better wall contact
– In an early superior or inferior location with good visible wall contact, note “reference impedance” for comparison with future positions
– Stable impedance over a respiratory cycle (if the change is ≤15-20 ohms) may indicate consistent wall contact
– Abnormally high impedance may indicate electrode is in a side branch
.png)
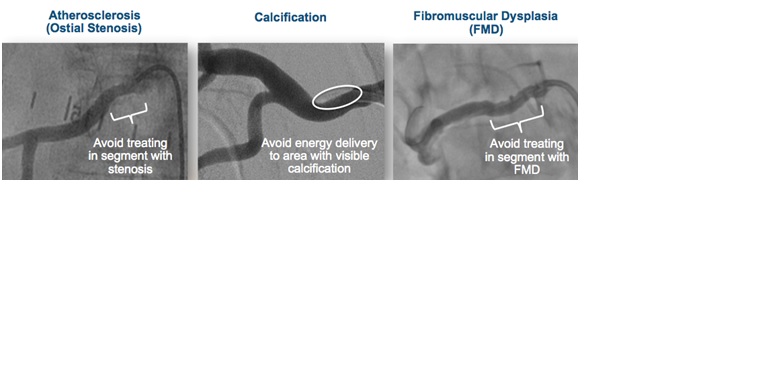
Areas to Avoid
– Avoid placing the electrode at the ostium of small branch arteries, such as those perfusing the adrenal glands
– There is no clinical experience treating near any areas of visible atherosclerosis, calcification, or fibromuscular dysplasia. Avoid treating areas of visible disease.
– There is no clinical experience treating in vessels with renal artery aneurysms
– The proprietary generator algorithm will occasionally stop RF delivery if certain temperature and/or impedance thresholds are crossed
– In these cases, some messaging will be displayed to guide users on how to proceed
– Check status messages can be cleared by pressing the foot switch twice within 3 seconds or by pressing the “continue” softkey on the generator

Appropriate Renal Denervation Patient Selection Requires a Systematic Approach


Symplicity RDN Global Clinical Program

Symplicity HTN-1
First-in-Man Cohort:
• 45 patients, EU, Australia
• Non-Randomised
• First patient enrolled: June 2007
• 12-month initial report in The Lancet, 2009
• Expanded Cohort 153 patients, EU, Australia, USA
Non-Randomised 36-month follow-up

Symplicity HTN-1 Patients Reflect Treatment Resistant Population

Symplicity HTN-1: Significant ,Sustained BP Reduction 3 years






